-
 Laboratory for arterial hypertension and hypertensive end-organ damage
Laboratory for arterial hypertension and hypertensive end-organ damageUlrich Wenzel
Mission Statement
“Understanding the underlying mechanisms for better prevention and treatment of arterial hypertension and hypertension induced end-organ damage”
— Ulrich Wenzel, MD
Team Members

Nicolas Ehnert

Stefan Gatzemeier

Leonie Dreher, MD
Research
Increasing evidence indicates that hypertension and hypertensive end-organ damage are not only mediated by hemodynamic injury. Inflammation plays also an important role in the pathophysiology and contributes to the deleterious consequences of arterial hypertension. Sodium intake is indispensable for normal body function but can be detrimental when taken in excess of dietary requirements. Recent data show that sodium levels also modulate the function of monocyte/macrophages, dendritic cells, and different T cell subsets. Some of these effects are mediated by changes in the microbiome and metabolome that can be found after high salt intake.
The purpose of our work is to discover the mechanisms underlying arterial hypertension to better understand arterial hypertension and the resulting end-organ damage. We focus on three points:
- Role of the complement system in hypertension.
- Role of aldosterone and the mineralocorticoid receptor in inflammatory cells.
- Pathogenesis of so-called malignant hypertension and malignant nephrosclerosis in humans.
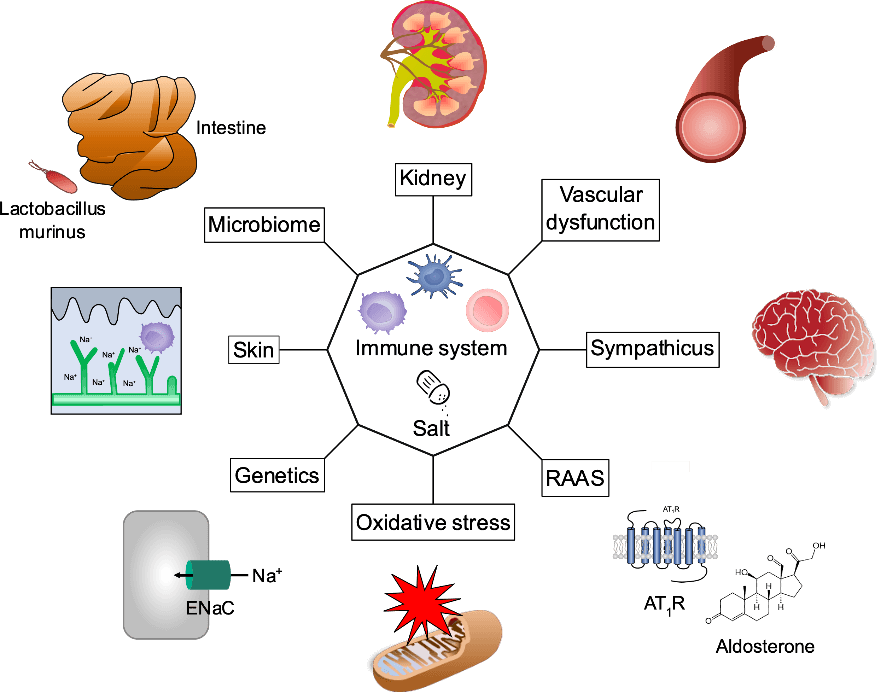
Figure 1: Our current concept explaining arterial hypertension (from Hengel et al. 2022). We call it a modified mosaic theory putting inflammation and salt in the center.
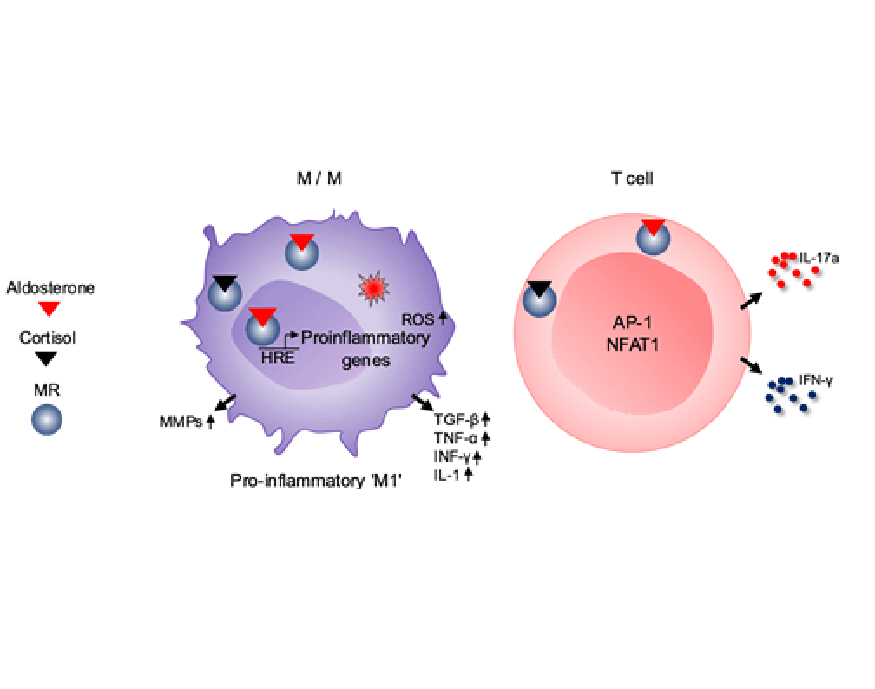
Figure 2: Proinflamamtory effects of the mineralocorticoid receptor in innate and adapive immune cells (from Hengel et al 2022). Recent data suggest that the mineralocorticoid receptor is expressed in inflammatory cells and has proinflammatory effects.
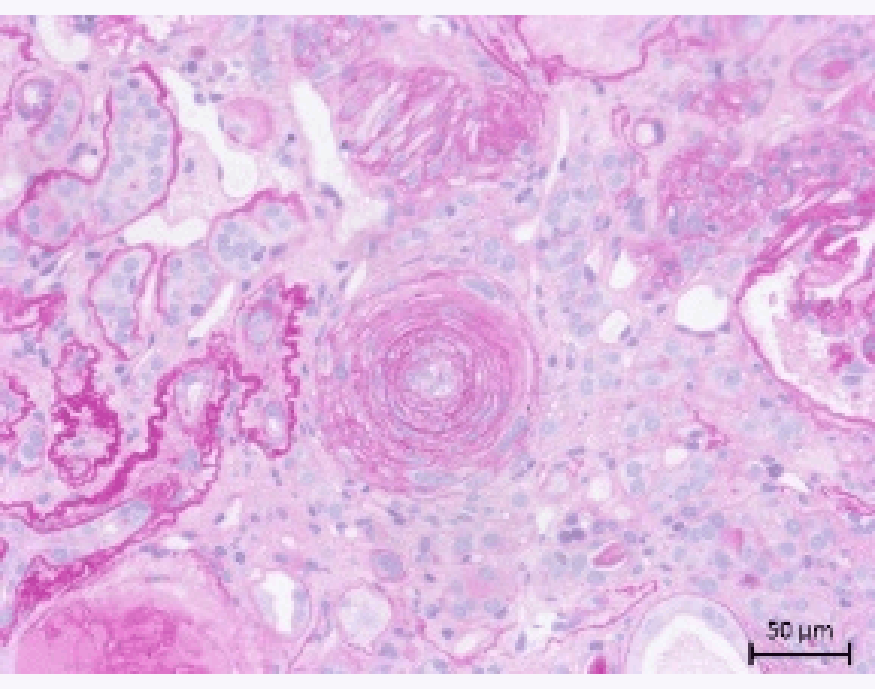
Figure 3: The pathogensis of renal injury in so called malignannt nephrosclerosis is not understood. It is a form of thrombotic microangiopathy defined by progressive narrowing of interlobular arterial branches and afferent arterioles with intima fibrosis and onion-like appearance of intimal scarring as shown below (histology provided by T. Wiech).
©Ulrich Wenzel

Ulrich Wenzel, MD
Senior Physician
III. Department of Medicine, University Medical Center Hamburg-Eppendorf (UKE)
Martinistr. 52
20246 Hamburg, Germany
CV
Senior Physician
|
1983 - 1989
|
Medical School, Freie Universität Berlin, Germany |
|
2002 - 2020
|
Habilitation and Venia legendi Internal Medicine, University Medical Center Hamburg Eppendorf, UKE |
|
1992
|
Medical Thesis, Freie Universität Berlin |
Clinical postgraduate education:
|
2005
|
Hypertensiologe DHL® |
|
2004
|
Board Certification Nephrology |
|
2000
|
Board Certification Internal Medicine |
Scientific postgraduate education:
|
1993 - 1995
|
Post-Doc, University of Texas, Health Science Center at San Antonio, Department of Medicine, Division of Nephrology (H. Abboud, M. D., Professor of Medicine), San Antonio, Texas, USA |
|
2020 - 2023 - 2020
|
President German Society of Hypertension |
|
2016
|
Franz Gross Award, Germany Society of Hypertension |
|
2003
|
Dr. Martini-Award, Hamburg, Germany |
|
2001
|
Adalbert Buding Award, Germany Society of Hypertension |
|
1988 - 1989
|
Fellow Studienstiftung des Deutschen Volkes |
Selected publications
1. | Bode M, Herrnstadt GR, Dreher L, Ehnert N, Kirkerup P, Lindenmeyer MT, Meyer-Schwesinger CF, Ehmke H, Köhl J, Huber TB, Krebs CF, Steinmetz OM, Wiech T, Wenzel UO. Hypertension. 2024 Jan;81(1):138-150. |
2. | Bode M, Diemer JN, Luu TV, Ehnert N, Teigeler T, Wiech T, Lindenmeyer MT, Herrnstadt GR, Bülow J, Huber TB, Tomas NM, Wenzel UO. Br J Pharmacol. 2023, 180:2412-2435. |
3. | The role of the mineralocorticoid receptor in immune cells in cardiovascular disease. |
4. | Mosaic theory revisited: inflammation and salt play a central role in arterial hypertension. |
5. | The role of complement in arterial hypertension and hypertensive end organ damage.. |
6. | The chemokine receptor CX3CR1 reduces renal injury in mice with angiotensin II-induced hypertension. |
7. | Adaptive immunity and IL-17A in chronic kidney disease in mice. |
8. | Salt, inflammation, IL-17 and hypertension. |
9. | The complement receptor C5aR1 contributes to renal damage but protects the heart in angiotensin II-induced hypertension. |
10. | Immune Mechanisms in Arterial Hypertension. |
News

Immune Response to Malaria and Chagas disease
PD Dr. Thomas Jacobs, Bernhard Nocht Institute for Tropical Medicine, „Immune Response to Infection“ ...
Martinistraße 52
Campus Research N27
20246 Hamburg Germany




